Consequently, other factors like the documentation of the clinical data, which has not been carried out precisely enough in the TDM process, could have had an impact.

Their model was least biased and most precise despite the fact that other models had bigger datasets. (2016) reveal differences in the accuracy and precision of pharmacokinetic models based on different data bases. analyzed the influence of erroneous patient records on population pharmacokinetic modeling and individual Bayesian estimation and drew attention to a more conscious documentation of sampling times ( Van Der Meer et al., 2012). Indeed, uncertainties of a few minutes are considered as a minor deviation and are deliberately accepted. In a daily clinical setting, it is often the case, that the planned sampling times are recorded rather than actual times, without being fully aware of the exact implications this may have. Apart from developing dosing recommendations in these populations, PK models derived from clinical routine data are used for model-informed precision dosing to calculate the optimal dose in combination with individually measured patient characteristics ( Wicha et al., 2015 Broeker et al., 2019). Routine clinical therapeutic drug monitoring (TDM) data is often used to develop population pharmacokinetic (PK) models, in particular for special patient populations ( Broeker et al., 2018). Thus, our results underline the importance of accurate documentation of time. falsely overestimated time above MIC (T > MIC) when true T > MIC was <0.4 and thus patients at risk of undertreatment), while the accurate model gave precise estimates of the indices across all simulated patients.Ĭonclusions: Even 5-minute-uncertainties caused bias and significant imprecision of primary population and individual PK parameters. The misspecified model provided biased PK/PD target information (e.g. Meropenem as a short half-life drug (~1 h) was more affected than caspofungin (~ 9–11 h). On the individual level, the rBias of the planned scenario for the typical values V1, Q and V2 increased with increasing uncertainty in time, while CL, AUC and elimination half-life were least affected. Results: On the population level, the estimates of the proportional residual error (prop.-err.) and the interindividual variability (IIV) on the central volume of distribution (V1) were most affected by erroneous records in the sampling and infusion time (e.g. The accurate and the misspecified (using planned sampling times) model were used for Bayesian forecasting of meropenem to assess the impact on PK/PD target calculations relevant to dosing decisions. Relative bias (rBias) and root mean squared error (rRMSE) were calculated to determine accuracy and precision of the primary and secondary PK parameters on the population and individual level. The estimation step was then performed with the accurate or planned times (replacing real time points by scheduled study values). deviation between accurate and planned sampling and infusion times (standard deviation (SD) ± 5 min to ± 30 min) were added randomly in R before carrying out the simulation step.
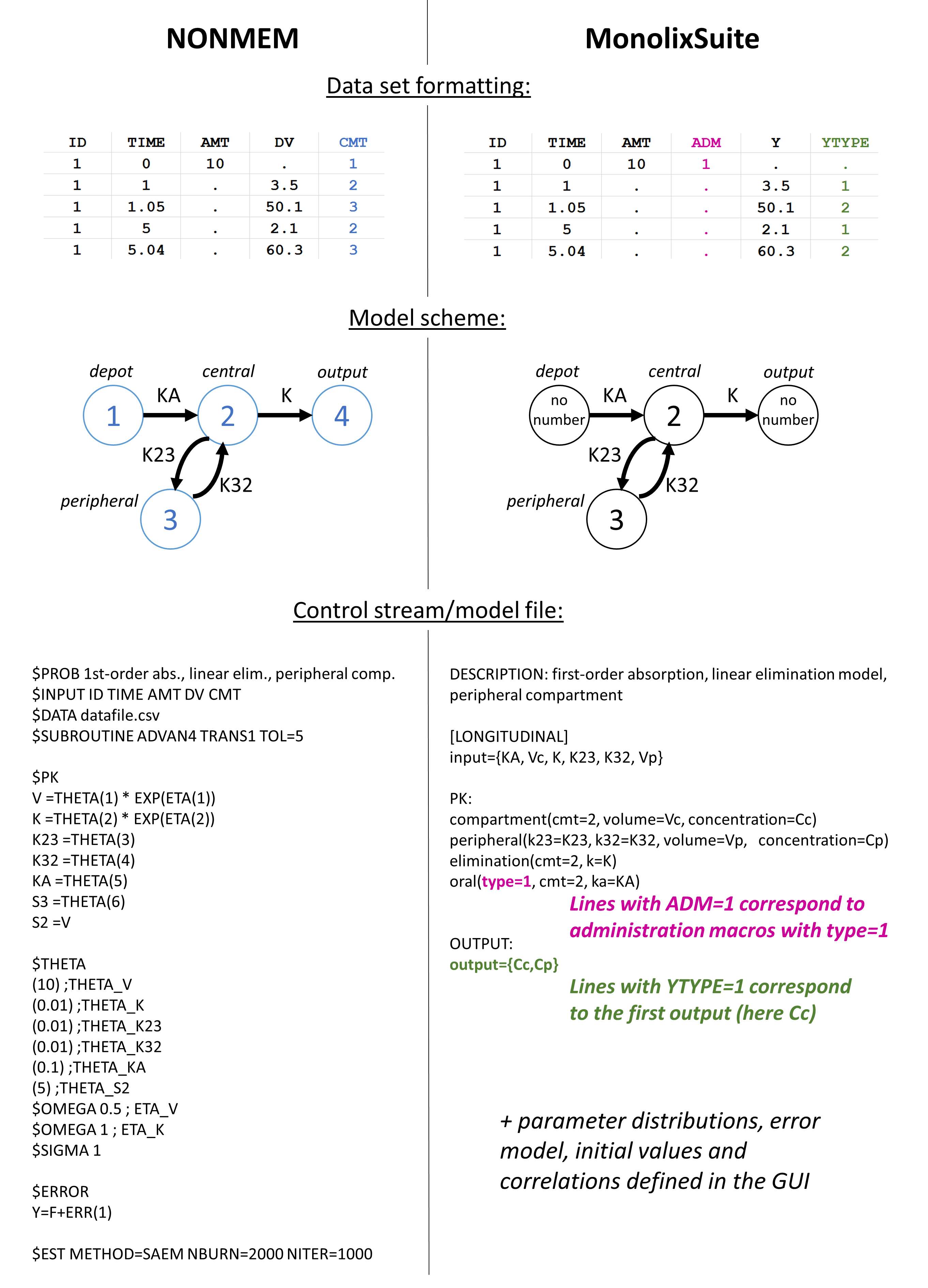
Methods: A stochastic simulation and estimation study was performed in NONMEM ® using previously published population PK models of meropenem and caspofungin. The aim of this study was to investigate uncertain documentation of (i) sampling times and (ii) infusion rate exemplified with two anti-infectives. The impact of uncertainty in documented sampling and infusion times in population PK modeling and model-informed precision dosing have not yet been systematically evaluated.

